InVivo Biosystems Blog

Parkinson’s Takes Decades to Develop–Zebrafish Provides Answers Within Days.

Unraveling Epilepsy: Modeling Dravet Syndrome in Zebrafish

Speeding up the diagnostic journey for Rare Disease patients: An in vivo discovery platform that helps classify variants of uncertain significance

New drugs, No Animals, No Problem. The FDA announced legislative changes – drugs are no longer required to be tested in animals before moving to human trials. What does that mean?
By:
Whole-gene Humanized Animal Models (WHAM) with CRISPR – Re-imagining how Rare Disease therapeutics are developed

Using Zebrafish Custom Injection Mixes to Accelerate the Grant Application Process
By:

WHAMyness – How Gene-Humanized Animals Can Dramatically Improve Drug Discovery

America is finally investing in Biotechnology & Biomanufacturing in a Big Way
By:

The Path to Affordable Therapeutics in Rare Disease – Tackling Congenital Disorder of Glycosylation in the PMM2 gene [Customer Story].
[Patent Announcement] InVivo Biosystems has officially been issued a patent on WHAM-humanization in the nematode, C. elegans
By:

Looking at Gene Paralogs across both zebrafish and C. elegans Model Organisms [Customer Story]
By:

Chemicals Models of Parkinson’s Disease in Larval Zebrafish
Poking its way into Western Medicine: How Acupuncture Could Help Ease Chronic Pain
By:

A Natural Fit – Using Zebrafish to Evaluate Natural Compounds for Anti-Inflammatory Properties

Quick Guide to Natural Products: What They are and How They Impact You
By:
Neuroscience Models: Transgenic Mice, Zebrafish, C. Elegans – And Jellyfish?
By:

From ED to AD: a drug repurposing success? How a performance-enhancing drug is giving insight into dementia
By:

A Closer Look at Nutraceutical Regulations: How Does the USA Compare?
By:

Testing the Neuroprotective Effect of a Lead Compound Using C. elegans for Fast and Cost-Effective Answers [Customer Story]

HIV, COVID-19, Alzheimer’s: The Impact of The FDA’s Expedited Programs and What’s Next
By:

How Can a Drug Get to Market Faster? Explaining the FDA’s Expedited Drug Tracks
By:

Let’s Fail Faster: Why Scientific Failures Should be Accepted as Progress
By:

The DrugAge Database: Showcasing C. Elegans In Longevity Research
Drug Repurposing for Neurological Disorders
By:
Neurological disorders are extremely widespread, affecting over 1 billion people worldwide, and have debilitating effects, impacting the brain, spinal cord, cranial nerves, and peripheral nerves.

Whole Gene Humanization for Personalized Medicine Applications
By:
We have found whole gene humanization can create a platform for highly translatable results in a model organism. Currently there is a big need for

How to Design a Good Phenotype Based Drug Discovery Program, and Why It Matters.
By:
With the advent of the molecular biology revolution and the human genome project in the 1990s there was a historic shift toward target-based drug
Understanding GPCRs Using Humanized C. elegans
By:
Our CSO, Dr. Chris Hopkins presented at DR. GPCR SUMMIT 2020 on why C. elegans is an ideal model to study the underlying principles of GPCRs.

Longevity Studies Using C. elegans to Identify Potential Anti-Aging Compounds [Customer Story]
By:
Dr. Vollmer shares his experience working with us and how the Longevity Platform delivers the results he needs within his timeline and budget.

Seventeen Minutes of Science: Viral Gene Therapies and Their Potential Use Against COVID-19
By:
Tune in weekly to our virtual series “Seventeen Minutes of Science” every Tuesday at 11am PST / 2pm ET where we go live on Facebook

Zebrafish and C. elegans in ALS: tightening the bench-to-clinic gap
By:
While advances in molecular imaging, high-throughput screening, genomics and techniques like CRISPR-Cas9 gene editing have dramatically enhanced our understanding of the human body and disease,
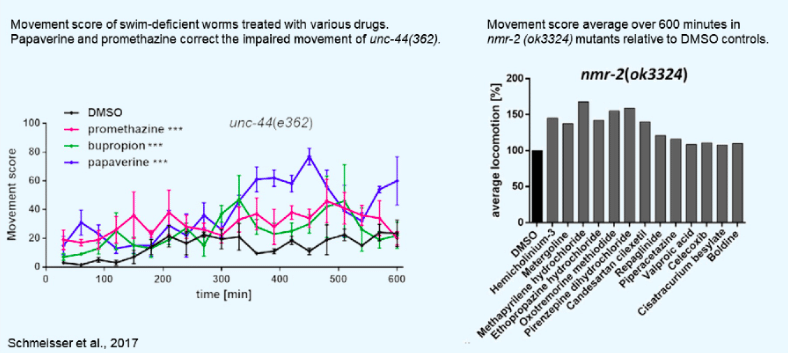
wMicroTracker Applications: Drug Screening
By:
The wMicroTracker is a great product to add to your drug screening toolkit. Check out the application sheet below to see how easy it is
Load More