Summary
Zebrafish models largely began as a way to research developmental biology, however, their potential to be a preclinical model of human disease quickly became apparent. While the pharmaceutical industry initially invested in zebrafish models, they have yet to widely adopt it – it’s time for that to change.
George Stresinger began using zebrafish as a model in his lab at the University of Oregon in the 1960s, and the model’s notable advantages drew interest from his colleagues in academia. Today zebrafish are a well-established and widely used model in the academic research community, but they have a rockier history for pharmaceutical research and therefore they have not gained as much popularity within this industry.
In the early 2000s the pharmaceutical industry, having taken note of the zebrafish model’s successes within academia, began using larval zebrafish assays to test drug safety and identify toxicity in compounds. Companies such as AstraZeneca, Bristol-Myers Squibb, Novartis, and Pfizer created in-house zebrafish facilities, and even more companies collaborated with academics or CROs (contract research organizations) to conduct research [Table 1] (Fleming & Alderton, 2013). The pharmaceutical industry’s commitment to zebrafish seemed strong: from 2006 to 2013, 24 zebrafish papers were published with co-authors from pharmaceutical companies, and a survey by the Safety Pharmacology Society found that 59% of respondents were actively investing in zebrafish assays with a focus on research into the CNS, and cardiovascular, auditory and visual systems (Fleming & Alderton, 2013).
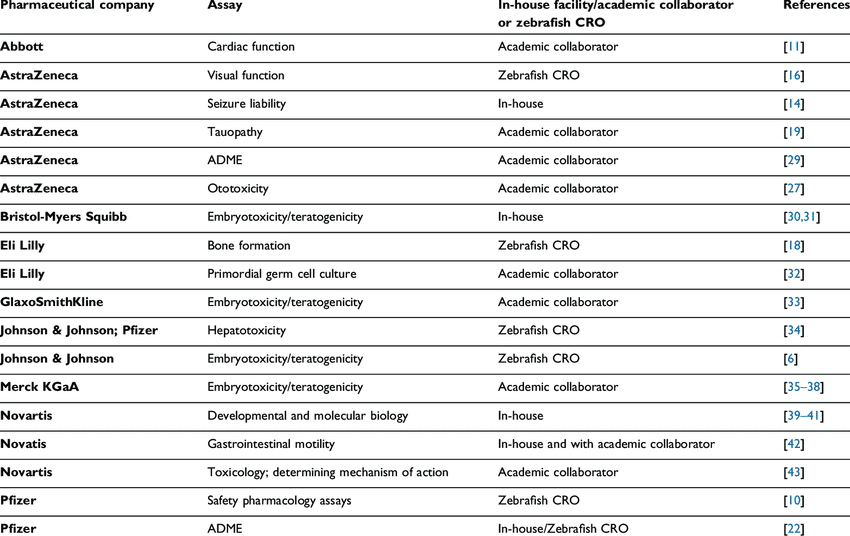
Table 1. Publications by pharmaceutical companies using larval zebrafish assays (2006 – 2012) (Fleming & Alderton, 2013).
However, pharmaceutical zebrafish research had a lull period, from around 2012 – 2015, as researchers encountered the practical complications of conducting drug screens in the newer zebrafish model (new compared to a traditional mammalian model) [Figure 1] (Patton, Zon & Langenau, 2021; Miyawaki, 2020). For instance, libraries and methodologies weren’t yet as well-established in the zebrafish model as they were in the historically more popular models such as cells and rodents.
In the last few years, however, there has been a reemergence of the zebrafish model in pharma research, with many researchers speaking out about their advantages, specifically for application in the drug discovery field. Networks such as Zfin and the Zebrafish Disease Models Society have worked to make large databases publicly available to researchers, and the rapid advancement of technology has made zebrafish a prime candidate for disease modeling, drug screening, target identification, and toxicology (MacRRae & Peterson, 2015). These advancements, paired with the pharmaceutical industry’s pressing need for lower cost, more robust models mean that there has never been a better time for zebrafish to be widely adopted by the field.
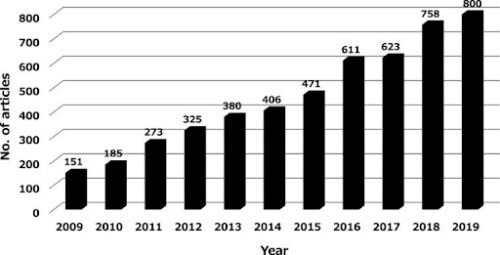
Figure 1. Number of research articles which include terms ‘zebrafish’ AND ‘toxicity’ OR ‘toxicology’ in Pubmed from 2009-2019 (Miyawaki, 2020).
The Need for a faster and more cost effective way to find & test compounds
Drug development is notoriously expensive and laborious: on average, a drug takes 7-10 years and $500 million to develop (Ogbru, 2020). With drug prices skyrocketing over the past few decades, the pharma industry needs a better way to develop therapies, as this is clearly not sustainable for neither the industry nor the consumer.
Novel high throughput in vivo systems are needed for early stage drug screening.
To cut costs and time, both in vivo and in vitro studies are used during the drug development process. However, while in vitro studies offer time and cost-cutting advantages, they have the reputation of being less translatable to humans. Indeed, on average, only five out of 250 compounds selected from in vitro drug discovery programs for pre-clinical testing will enter clinical trials (Tang, 2017; Fleming & Alderton, 2013). Conversely, In vivo testing has the advantage of capturing the impact of a compound on the whole organism, rather than how cells react in isolation, but is typically more expensive, time consuming, and requires compliance with the 3Rs.
How zebrafish fulfill these needs:
Zebrafish are an extremely useful model for pharmaceutical research as they are a first-rate laboratory animal: easily maintained, cost effective to house, have a large brood size(200 eggs per week), are amenable to genetic manipulation, and have 70% of genomic homology to humans (84% of known human disease-causing genes) [Figure 1]. Also, as a vertebrate model, zebrafish are not covered by the 3Rs, and thus, do not require the amount of regulatory paperwork that their mammalian counterparts do.
Furthermore, zebrafish have specific advantages for toxicology research such as their short lifetime which makes it easy to study disease or development, and their external fertilization which provides easy access to embryos. Furthermore, zebrafish are easy to test compounds on since zebrafish can absorb compounds through their mouth, gills, or skin or by direct injection into the yolk sac, vein sinus, or circulating blood. Zebrafish also enable researchers to perform in vivo assays, which are are important for drug discovery, as they enable toxicity to be assessed more reliably in a multi-organ organism, unlike an in vitro assay which can only detect tissue-specific toxicity (Miyawaki, 2020).
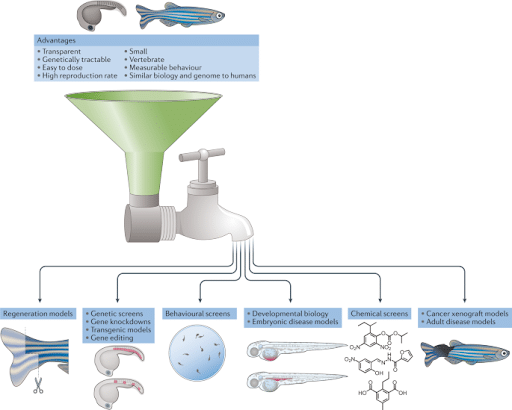
Figure 2. Zebrafish can model human disease and drug treatments in the embryonic and adult stages. During these embryonic stages, the biology and function of organs and tissues can be followed in living animals at single-cell resolution to study diverse and complex processes including developmental biology, developmental disease models, stem cells and regeneration, and neural circuitry and behaviours. By 3 months, zebrafish are breeding adults and generate large numbers of embryos for small-molecule and genetic screens, and are themselves studied as models for disease, including cancer, cardiovascular and other organ defects, behaviour, addiction, metabolism, and adult tissue regeneration and physiology (Patton, Zon & Langenau, 2021).
Recent advances that have refined zebrafish’s utility as a whole-animal model for preclinical drug repurposing & drug development
Between the rapid progression of technology, and the growth of the zebrafish community, new models, databases, and tools have been developed. Some which particularly benefit zebrafish’s application in the pharma industry include:
- Generation of transgenic zebrafish models
- Creation of databases such as Zfin which provide publicly available information on the husbandry and handling of zebrafish as well as information on fish lines
- Allograft and xenograft transplant assays (can use both larval and adult fish) enable the assessment of drug effects in live animals and directly within human cancers
- Ability to conduct high-throughput screens
Applying zebrafish to today’s pharmaceutical industry
The tide is beginning to change for zebrafish, as a recent article noted that zebrafish are making their way into pharmaceutical companies’ research again [Table 2] (Cully, 2019).
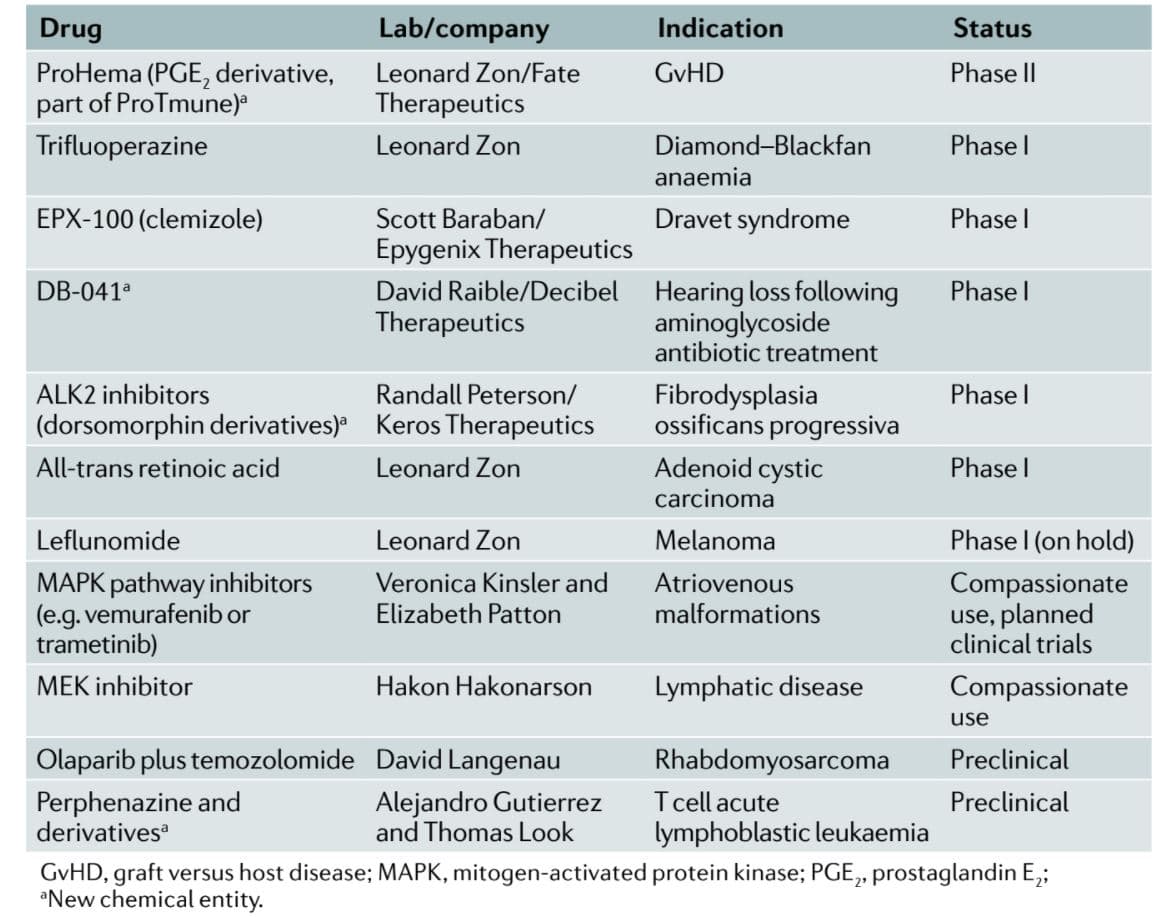
Table 2. Nearly ten compounds that were identified in zebrafish screens are in or about to enter clinical trials (Cully, 2019).
The increase usage of the zebrafish model by pharma companies is particularly noteworthy for neurological and CNS research, as Dr Peterson, the Dean Pharmacy at the University of Utah, explained, “a lot of these big pharmaceutical companies [are] getting out of neuroscience altogether at a time when that’s probably where the greatest medical need is, because it’s just so daunting to use conventional tools” (Cully, 2019).
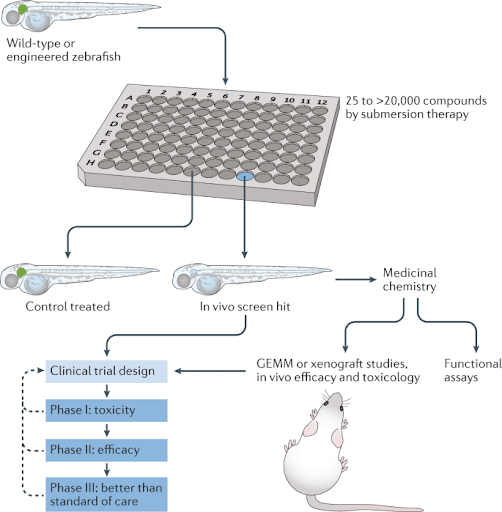
Figure 3. Zebrafish are innovative drivers of drug discovery and drug-lead optimization in the drug development pipeline.
While zebrafish have exciting potential, they also have some notable limitations such as their small size, which requires technical skills for experimental operations (administration and blood/tissue sampling in adult zebrafish, among others). This is where collaborating and outsourcing can be extremely beneficial. By utilizing the expertise of another lab (either an academic or CRO lab), pharmaceutical companies can relieve themselves of the burden of the operational side of using an animal model, and instead, streamline their drug discovery process.
Recently, Forbes published an article on the booming CRO market (expected to grow by 12% in 2022, and reach $71.7 billion by 2024) (Lucas, 2021). This article emphasized the role CROs can play in the pharmaceutical industry – providing cost and workforce saving way to bring drugs to the market faster. Unlock cutting-edge zebrafish research solutions now with Invivo biosystems – your premier zebrafish CRO partner.
To check out InVivo Biosystems’ offerings click here. Or reach out to one of our experts today.
References
- Cully, M. (2019). Zebrafish earn their drug discovery stripes. Nature reviews, drug discovery. https://www.nature.com/articles/d41573-019-00165-x
- Fleming, A. & Alderton, W.K. (2012). Zebrafish in pharmaceutical industry research: Finding the best fit, Drug Discovery Today Disease Models 10(1):e43-e50 , DOI:10.1016/j.ddmod.2012.02.006
- Lucas, Nick (2021). Contract Research Organizations: Key Partners in the Drug Development Journey. https://www.forbes.com/sites/forbestechcouncil/2021/04/09/contract-research-organizations-key-partners-in-the-drug-development-journey/
- MacRae, C. A., & Peterson, R. T. (2015). Zebrafish as tools for drug discovery. Nature reviews. Drug discovery, 14(10), 721-731. https://doi.org/10.1038/nrd4627
- Miyawaki I. (2020). Application of zebrafish to safety evaluation in drug discovery. Journal of toxicologic pathology, 33(4), 197-210. https://doi.org/10.1293/tox.2020-0021
- Patton, E.E., Zon, L.I. & Langenau, D.M. Zebrafish disease models in drug discovery: from preclinical modelling to clinical trials. Nat Rev Drug Discov 20, 611-628 (2021). https://doi.org/10.1038/s41573-021-00210-8
- Tang, Candice (2017). In vitro vs. In vivo: Is One Better? University Health Network, https://www.uhnresearch.ca/news/vitro-vs-vivo-one-better
Take a step towards more ethical and efficient toxicity testing with our zebrafish toxicity testing service. By leveraging the remarkable attributes of zebrafish as a model organism, we offer a cutting-edge solution for evaluating the safety of chemicals and substances.
Take the leap into the future of drug discovery. Contact us now to unlock the potential of our comprehensive drug discovery services and accelerate the development of life-changing therapies.



