Summary
Rising healthcare costs mean it’s time for pharmaceutical companies to use non-mammalian models in their drug development process – by saving time and money pharmaceutical companies can make a return on their products without sky-high prices for consumers.
It is no surprise that healthcare costs are rising, but the figures are staggering: it is reported that healthcare spending in the USA increased, “nearly a trillion dollars between 2009 and 2019, when adjusted for inflation, with service price and intensity, including the rising cost of pharmaceutical drugs, made up more than 50% of the increase” (Peterson Foundation, 2021). With seniors driving the healthcare spending, and the country having an aging population, (currently 16.5%, of the total population but expected to exceed 20% by 2030), this creates a concerning trend (Census Bureau, 2020).
The number of people enrolled in Medicare has tripled since 1970, climbing from 20 million in 1970 to 61 million in 2019, and it is projected to reach about 88 million in 30 years.” -Peterson Foundation (2020).
Since drug consumption fuels a large portion of healthcare costs, one way to lower the overall cost of healthcare is by making drug development more economical. Currently, getting a drug to market is extremely time-consuming and expensive: taking anywhere from 7-10 years, and costing an average of $500 million per drug (Ogbru, 2020). When you take these costs of time and money into consideration, the pricing of pharmaceuticals can be understandable, even necessary. Especially considering that most of this money is spent in the early phases, and there are no guarantees: if the drug doesn’t work or it is not approved, then the company must absorb the costs. Even when drugs are approved they have to be marketed and studied further, as a result, only 3 out of every 20 drugs ever generates enough profit to cover the costs of their development.
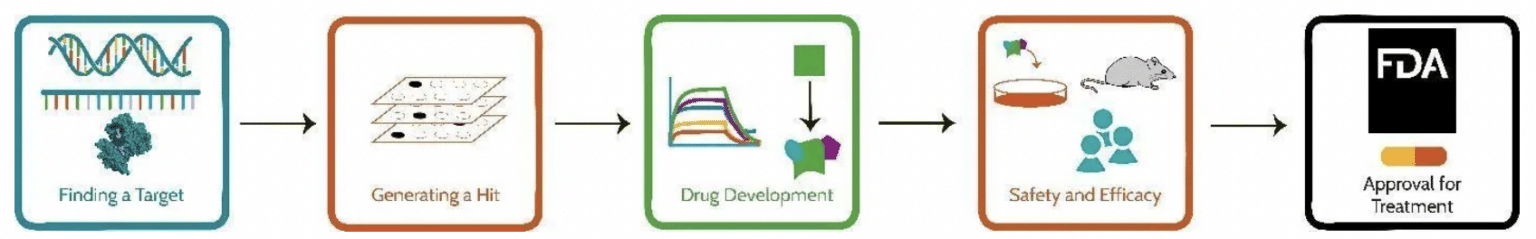
Figure 1.The process of discovery, from finding a target to FDA approval (Rowe, 2020. SITN).
But why is this the norm? Professor Daria Mochly-Rosen explained that, “when we’re making drugs, we have to be better than nature” (James, 2010). While this is true, we can also use nature to our advantage: for instance, in the selection of our model organisms. Historically, mammalian models have been used in drug development (by some estimates, mice are still used in up to 90% of pre-clinical trials), however, incorporating non-mammalian models in the early phases of drug development can significantly decrease the time and money that these phases take (Vitale, 2019). If pharmaceutical companies can save their resources in this way, they can then price their drugs at a lower cost, and in turn consumers (healthcare patients) can also see their costs decrease.
For example, take a drug that is developed using an invertebrate model instead of a mouse model: invertebrates’ are advantageous as their brief life cycle means longitudinal studies can be completed in a shorter time period, and their small size means that they require less space for housing, both of which result in lower costs. Furthermore, there are fewer ethical considerations when using invertebrates in studies compared to traditional mammalian models (Wilson-Sanders, 2011).
As the USA’s population ages, the strain on our healthcare system is growing as well. There are many ways we can combat this, but one of the most impactful can be the switch to a multi-model approach to drug development. Non-mammalian models such as C. elegans and Drosophila are already extensively used model organisms, and proven to be effective in drug discovery. However, mice are still considered the gold standard of model organisms. The next few decades should be the time to shift this usage – making animal models a later step in preclinical development.
References
- Census Bureau, (2020). Older Americans Month: May 2021. https://www.census.gov/newsroom/stories/older-americans-month.html
- James, Julia (2010). Why drug development is time consuming and expensive (hint: it’s hard). Scope, published by Stanford Medicine. https://scopeblog.stanford.edu/2010/07/21/mochly_rosen_lecture/
- Ogbru, Omudhome (2020). Why Drugs Cost So Much. MedicineNet. https://www.medicinenet.com/drugs_why_drugs_cost_so_much/views.htm
- Peterson, Peter G. (2020). Why are Americans Paying More For Healthcare? https://www.pgpf.org/blog/2020/04/why-are-americans-paying-more-for-healthcare
- Rowe, Sebastian (2020). Modern Drug Discovery: Why is the drug development pipeline full of expensive failures? SITN (Science in the News: a Graduate Student Group at the Harvard Graduate School of the Arts and Sciences). https://sitn.hms.harvard.edu/flash/2020/modern-drug-discovery-why-is-the-drug-development-pipeline-full-of-expensive-failures/#
- Vitale, James (2019). Animal Models in Drug Discovery. Taconic. https://www.taconic.com/taconic-insights/quality/animal-models-drug-discovery.html#footnote
- Wilson-Sanders, S.E. (2011). Invertebrate Models for Biomedical Research, Testing, and Education, ILAR Journal, 52 (2) 126-152, https://doi.org/10.1093/ilar.52.2.126
Discover the future of medicine through our comprehensive drug discovery development service. Reach out to us today and take the first step towards revolutionizing healthcare.



