Zebrafish Genome Editing Service
More Zebrafish CRO Services
Custom Zebrafish Genome-Editing Service for Precise Genetic Modifications
InVivo Biosystems is a leading zebrafish Contract Research Organization (CRO) specializing in zebrafish CRISPR knock-in and transposon insertion techniques and offers cutting-edge gene-editing services. Our repertoire includes mutagenesis, tagging, and transposon sequencing, leveraging CRISPR and Tol2 genome editing tools for zebrafish injection and zebrafish microinjection procedures.
As a licensed CRISPR solutions provider, our scientific team has 40 years of combined expertise in zebrafish gene editing, significantly streamlining your experimental design process. We cater to a variety of experimental needs, from gene function investigation through CRISPR-Cas9 gene editing to complex genetic modifications.
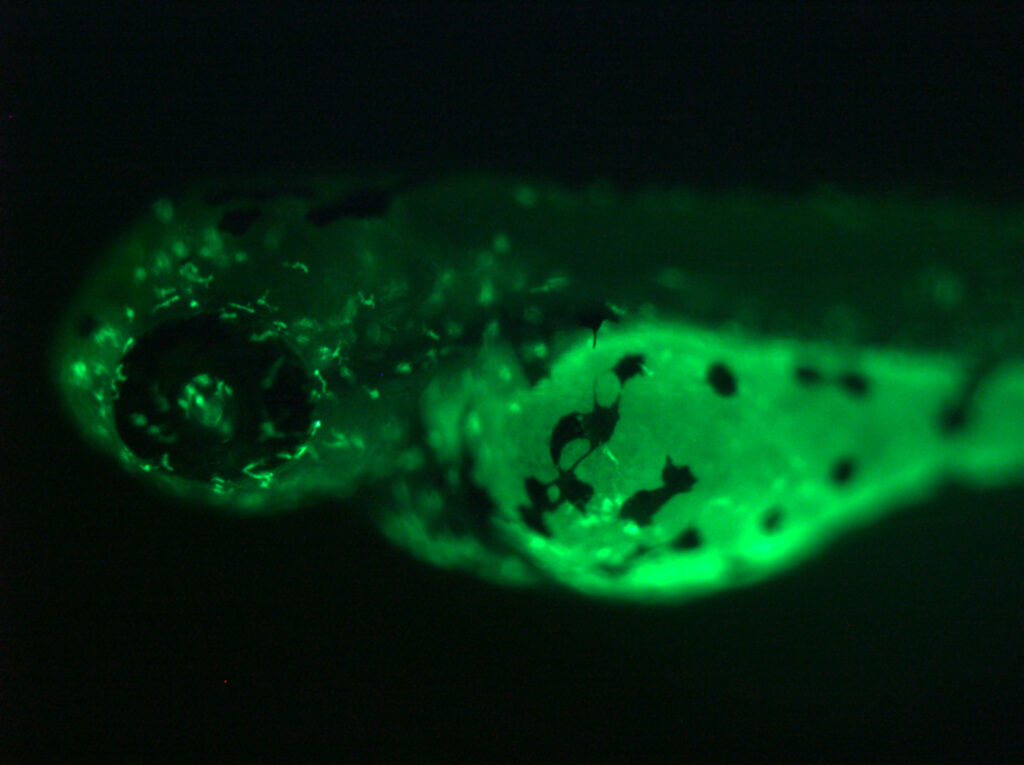
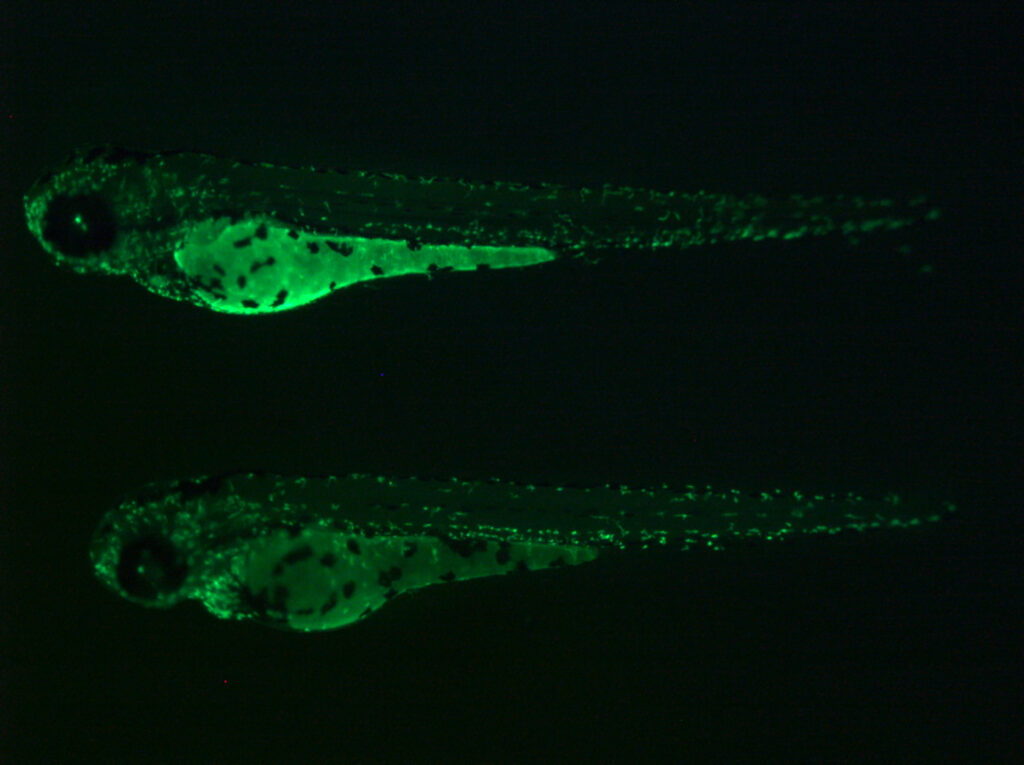
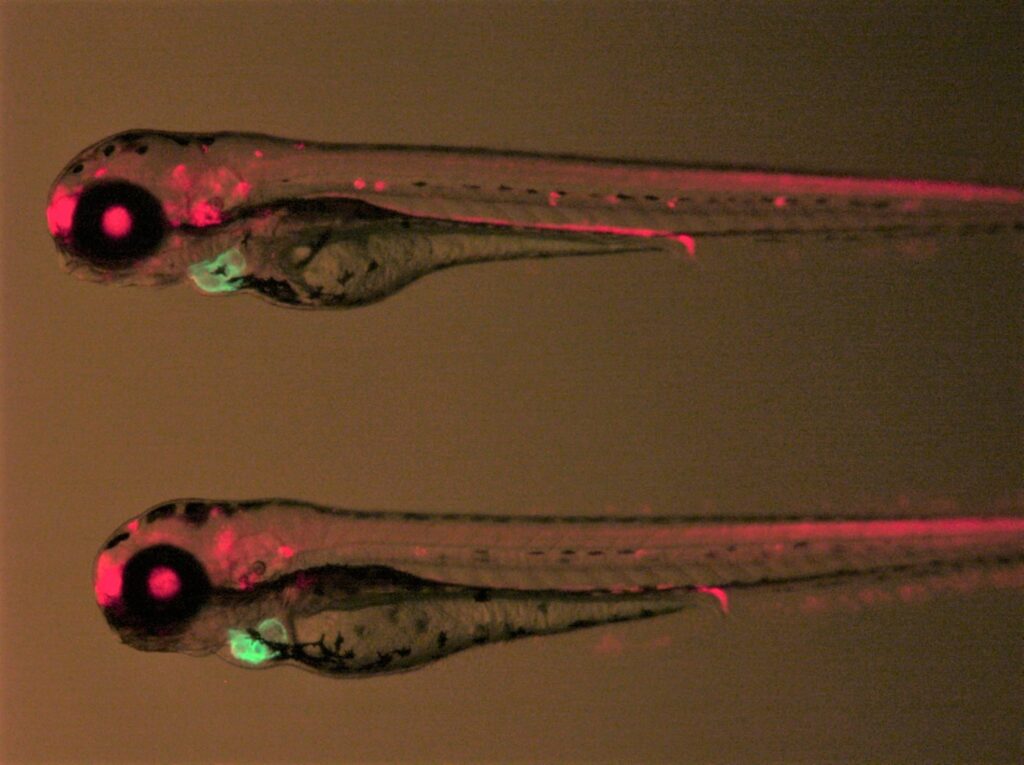
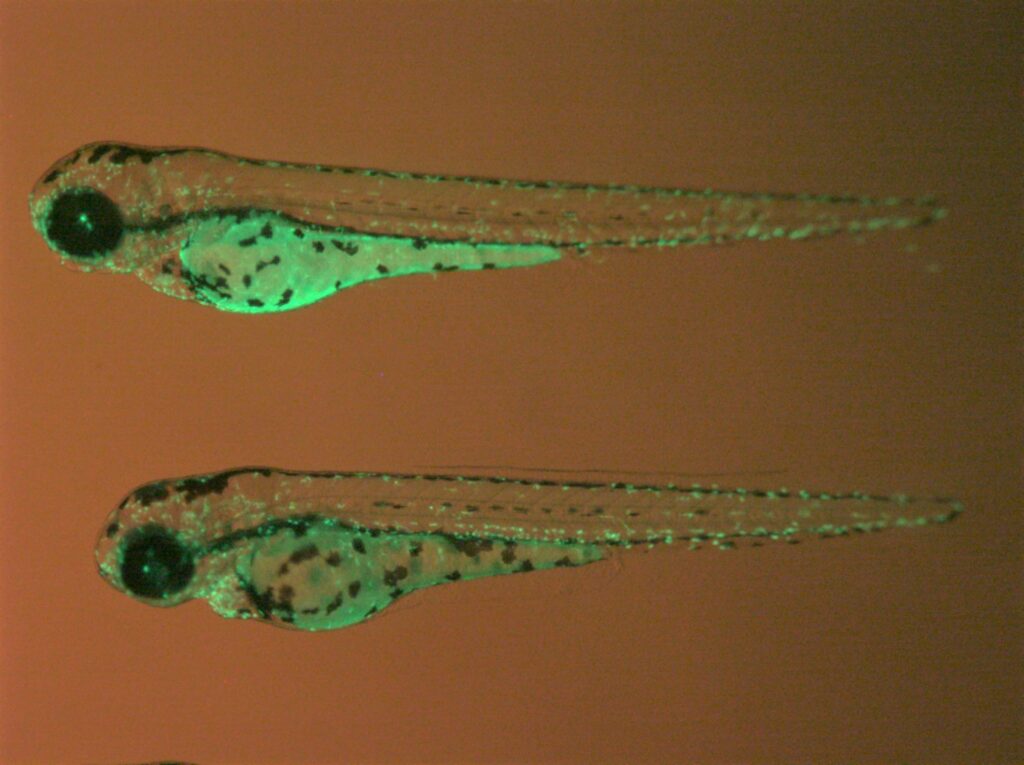
Zebrafish mfap4:EGFP transgenic lines. Green fluorescent labeling of macrophages built using our tol2 transgenic methods.

“Usually companies just give you the stuff and they don’t tell you how to screen it, they don’t offer the primers and you’re sort of stuck doing that on your own. [InVivo Biosystems] has the efficiency and the set-up to do these kinds of assays”
– Dr. Anita Quintana, Associate Professor, University of Texas at El Paso.

” I was able to use the injection mix to jump start my project by leveraging InVivo Biosystem’s design expertise to save time and get results more quickly.”
– Dr. Melissa Wright, Assistant Professor, Children’s Hospital Colorado Anschutz Medical Campus.
How To Choose Our Services
Our goal is to help you move forward with your experiments and accelerate the time from a project idea to the execution of the project. Select the service that best fits your needs, whether you’re new to zebrafish genome editing, an expert needing advanced support, or someone working with zebrafish but requiring additional help.
I am new to zebrafish genome editing
- I have an experimental idea, but not sure how to proceed
- I know the data I’d like to get, but am not sure what tool to use
- I’d like to talk to an expert
I am an expert in zebrafish gene editing
Explore our offerings.
I work with zebrafish, but need help
- Design
- Cloning
- Injection & In Vivo testing
- Identify a founder/Screening
- Selecting quality reagents
Types of Edits
We offer a variety of service packages to accommodate your lab’s needs and budget. Choices range from the fast, affordable Custom Injection Mix to the comprehensive Full Build service packages, allowing you to customize the support you need to accomplish your gene editing goals. Whether you’re interested in zebrafish knockout models for gene function studies or knock-in models for disease modeling, we offer precision, efficiency, and customization.
We work with you on project design using a collaborative and reliable approach to ensure that you receive the zebrafish line best suited for your experiments, whether it involves zebrafish knockout model building or exploring zebrafish functional genomics.
CRISPR Knockout
CRISPR Knock-in
Tol2
Service Packages
- We offer a variety of service packages to fit the needs and budget of your lab. Our services are fully customizable to get you the edit you want along with the support you need to start your project.
- CRISPR Injection Mix: Our Custom Injection Mix packages range from standard ready-to-inject mixes to full in vivo evaluation of sgRNA cutting efficiency and in-house validated screening tools to accommodate YOUR level of expertise. Learn more.
- Mosaic Clutch (F0 Injected Embryos): We develop and rigorously test your custom injection mix and deliver expertly injected embryos ready to submit to your nursery.
- Full Build (Sequence Verified Heterozygous Line): Our full service package handles the entire process of design, validation, injection, rearing, and germline screening, to deliver stable transgenic zebrafish lines.
The InVivo Biosystems Workflow
InVivo Biosystems provides a streamlined process for zebrafish gene editing services.

Project Design
Selection of best guide RNAs surrounding the locus to be edited.

sgRNA Testing
In vivo cutting assayed by PCR/sequencing to determine the most efficient sgRNA.

Donor Homology Design
Repair templates are designed around high quality sgRNAs.

Germline Verification
Verification of germline editing.

Clutch Verification
Evaluation of edit by PCR and/or sequencing of 10% of injected embryos.
Injection
Skilled injectors for rigorous in vivo testing of editing reagents.
Comparison of Zebrafish Genome Editing Methods
Nature of Edit
Number of Insertions
Germline Edits
Germline Efficiency
Common Terminology
Phenotype in F0 (injected embryos)
Max. Size of Insert
- Tol2 Transposase
- Random insertion
- Multiple
- Yes
- 10% to 40%
- Transgenesis
- Mosaic
- Usually 13-15kb - depends on cargo
- CRISPR/ Cas9
- Site specific
- Single
- Yes
- ~2% to 5%
- Knock-in Edit
- Mosaic
- Unknown but appears that the larger the insert the lower the efficiency.
- Morpholinos
- None
- n/a
- None
- n/a
- Knockdown
- Yes, but transient expression is variable, depends on quality of MO
- n/a
Note: testing your gene of interest (GOI) by comparing results from multiple methods can strengthen (or weaken) the interpretation of observed phenotype or importance of gene/mutation.
Tol2
Most commonly used in zebrafish labs to make fast and rapid genome edits by random insertion of reporter cassettes (e.g. GFP, overexpression of mutant proteins). However, this method is not locus specific. It does not always recapitulate native expression patterns of gene and does not produce disruption in WT sequence. InVivo Biosystems does not used the Tol2 method to produce mutants.
CRISPR/Cas9
Most commonly used to “Knock out” by INDEL (insertion/deletion around a double stranded break), “Knock in” point mutations (SDM; Site Directed Mutagensis) or Tags into native locus. A powerful genome editing method but it is more labor and time intensive to generate stable lines (germline). Compared to Tol2, CRISPR/Cas9 is less efficient but much more precise. F0 animals are called “Crispants”. These edits are highly mosaic in F0s which makes phenotyping challenging.
Morpholinos (MOs)
Most commonly used to get a functional “knockdown” of gene expression in F0 animals via small synthetic oligonucleotides that block translation or splicing of mRNAs. F0 animals called “Morphants”. Phenotyping is possible with good controls but declining concentration of MO leads to sub optimal knock down usually by 4dpf. Quick and very easy way to see if your GOI might have a phenotype and warrant making a germline mutant/null.
Our Capabilities
The InVivo Biosystems scientific team has 40 years of combined zebrafish gene editing experience. With access to the state-of-the-art University of Oregon Zebrafish Facility and world-renowned zebrafish researchers on campus, our team has successfully introduced point mutations by knocking in sequence variants to study gene function.
With our ability to generate zebrafish knock-in lines, we can:
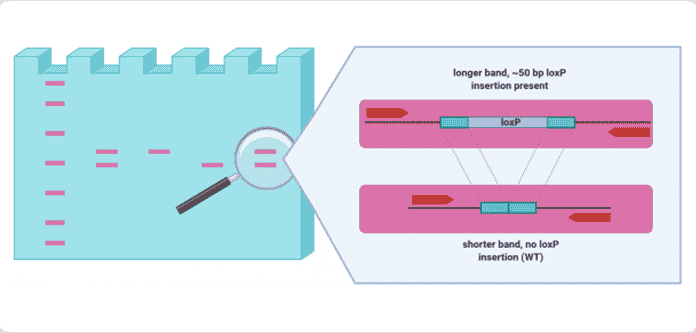
Schematic of a PCR gel testing 4 samples for a loxP insertion. When loxP is successfully inserted, DNA fragments in the amplification region are ~50bp longer. This difference can be seen on a gel, and founders transmitting this edit can be identified to establish a stable line. Schematic created using BioRender.
- Create Human Disease Models by introducing point mutations to model human genetic diseases.
- Track gene expression and protein localization by knocking in a fluorescent tag at the native locus.
- Add immunohistochemistry to your toolbox by knocking in a protein tag.
- Create conditional control of gene function by knocking in loxP sites.
FAQs
Frequently Asked Questions
Having very high similarity in genetic makeup with humans, zebrafish has proved to be an important model for human genetics and disease. By using zebrafish knockout or knock-in models, genes can be manipulated to understand different disease pathways.
Our scientific team has 40 years of combined zebrafish gene editing experience and access to the start-of-art University of Oregon Zebrafish Facility and top-rated zebrafish researchers. We use CRISPR and Tol2 as our genome editing methods. Our service can significantly reduce your experiment design time and can be tailored to different experimental requirements.
Complete Injection Mix is simple to work with, all you need to do is add water, inject, and screen. Our Complete CRISPR Injection Mix includes Cas9 protein, an RNA-based sgRNA(s), and a donor homology construct and can be shipped as a lyophilized mix in as little as 2 weeks.
Using the Tol2 transgenesis you can generate Cre Recombinase-expressing lines, transcription reporters, custom zebrafish models to analyze tissue-specific gene expression, models to control the timing of gene expression, and humanized zebrafish. Insertion content can range between 2 – 10 kb.
Using CRISPR/Cas9 technology, we can modify the zebrafish genome through Knockout, Point Mutation, Precise Deletion, Floxed Allele, etc. Specific experiment requirements can be met by working with our expert research design team members.
Prices stated are exclusive of all taxes, fees, licenses, duties or levies and, unless otherwise stated in the quotation, transportation charges, freight and insurance.
For Mosaic Clutch and Full Build services you will be billed on a 3 step payment plan: 30% to initiate Phase I, 30% to initiate Phase II, 40% is due upon project completion and delivery.
For all other project types (e.g., Custom Injection Mix, and evaluation of sgRNA cutting), full payment will be invoiced and due prior to shipment of the project, unless otherwise set forth in the Quote.
Live animal shipment depends on each individual country’s import regulations & guidelines. We can ship embryos or adult fish as long as they are allowed in the receiving country.
Every reasonable effort will be made to identify the desired germline edit. In the event the edit is not identified a husbandry charge will still be assessed. The client will not be liable for the remainder of the Phase III cost.
Yes, we do. In case you could not find what you are looking for. Please feel free to contact us at [email protected].
Key References
A well rounded review provides a detailed roadmap outlining -numerous tools available for genetic modification in zebrafish, including the historical and experimental context in which they have been used. Methods including Tol2 transgenesis, TALENs and CRISPR/Cas9 are discussed. A particularly useful flowchart can help readers understand the numerous decision points and considerations involved in choosing the right approach for their particular experimental needs.
An excellent framework for the novice and expert genome engineer alike.The article outlines the design of a number of elegant genetic tools to facilitate CRISPR/Cas9 mediated knock-ins, as well as downstream screening and experimental uses. The focus here is on fluorescent tags, epitope tags, conditional tools, and transgenesis markers to help researchers screen for phenotypically silent edits.
If you’re looking for more mechanistic insight into gene editing in zebrafish, look no further than this review from Simone et al. The authors provide a detailed discussion of the different DNA repair pathways, template types, and experimental considerations that all factor into a successful gene editing endeavor. The various “designer nucleases” available for zebrafish gene editing applications, their advantages, and their limitations are also discussed.



