Genome editing in Zebrafish
Genome editing refers to the technology that provides the capability of editing the organism’s native DNA sequence into a new and desired DNA sequence1. Scientifically speaking, zebrafish and mammalian genes are highly conserved, and around 70% of human disease genes have an ortholog in zebrafish23. By using genome editing, researchers nowadays are capable of studying gene function in new innovative ways, maneuvering its ability to precisely control the genome. At InVivo Biosystems, we specialize in multiple different ways of zebrafish genome editing such as an insertion, deletion, or replacement of DNA at a specific site in a genome to achieve site-directed mutagenesis, knock-in tagging, and Tol2 transgenesis. These all allow us use the zebrafish to study detailed characteristics of its human ortholog.
CRISPR genome editing
While CRISPR/Cas9 (clustered regularly interspaced short palindromic repeats/CRISPR-associated protein 9) technology is still relatively new (the first publication to mention it was in March of 2002), the technology has rapidly gained popularity and respect in the field. Just 18 years after the technology first appeared in print, the 2020 Nobel Prize in Chemistry was awarded to Emmanuelle Charpentier and Jennifer A. Doudna “for the development of a method for genome editing”2. Using CRISPR/cas9 technology that Charpentier and Doudna developed, researchers now are able to edit the DNA of animals, plants, and microorganisms with extremely high precision. This technology has had a revolutionary impact on the life sciences field by contributing to new cancer therapies and in the future, possibly in the treatment of several genetic inherited diseases. The emergence of CRISPR/Cas9 technology has revolutionized multiple scientific fields, including basic life sciences, agriculture, biodiversity conservation, biotechnology, COVID-19 testing24, etc..
CRISPR/Cas9 technology has been readily adopted in zebrafish research dating back to the early publications, such as Hwang et al., 2013. Most commonly used methods for genetic modifications to create stable transgenic lines including Tol2 transposon, and TALENs (transcription activator-like effector nucleases), predates the relatively new CRISPR/Cas9 technology. Specifically, in Zebrafish genome editing, CRISPR/cas9 systems mimic the original biological process adapted from bacteria and archaea, an adaptive immune response from degrading the viral molecules by using the tracRNA (CRISPR/cas) from Streptococcus pyogenes9 in degrading foreign DNA molecules. In a short period of time, many variations on CRISPR/Cas9 have emerged, including Cas12a, Cas12b, Cas12e, and Cas14 variants26, and continue to rapidly expand scientists’ gene-editing toolbox (figure 1).
In the zebrafish model system, the CRISPR/cas9 system has the ability to precisely edit the genome3. The editing process relies on the cell’s inherent capabilities of repairing the double-stranded breaks by at least three mechanisms4:
- Non-Homologous End Joining (NHEJ) mechanism,
- Homology Directed Repair (HDR),
- Microhomology-mediated end joining (MMEJ).
Non-Homologous End Joining (NHEJ) is the least precise DSB repair strategy. It is used in gene knock-out approaches by inducing a DSB without providing a template for repair with the possibility of introducing insertions and deletions during repair of the DSB5,9. Homology-directed repair (HDR), on the other hand, is the most precise repair mechanism. HDR uses longer sequences of complementary DNA as a template to replicate the lost sequence when a DSB occurs6,7,8,9. The third mechanism, microhomology-mediated end joining (MMEJ), can be utilized in specific gene editing without the inclusion of exogenous DNA if the induced DSB is targeted to a genomic location containing microhomology sequence repeats (nucleotides of sizes 3-30 bp that are complementary to each other at the DSB)7,9.
Advantages of using CRISPR/Cas9 system
The precision of CRISPR/Cas9 DNA editing makes it a valuable tool in zebrafish genome editing over other methods, such as Tol2 transposase which relies on random insertion9. For locus-specific CRISPR editing, the sgRNA (single guided RNA) injection that has been designed to recognize specific DNA sequences can be combined with an associated cas9 endonucleases. Guided by sgRNA, the Cas9 endonuclease breaks DNA at a targeted desired sequence. Imprecise repair of the double strand breaks can result in INDELs (insertion or deletion) mutations, while associated repair pathways can be engineered in such a way to introduce specific point mutations or insertions27. Moreover, natural repair mechanisms such as NHEJ, HDR, and MMEJ play a vital role in repairing the double-stranded breaks caused by the Cas9 + sgRNA leading to the precise zebrafish genome editing.
Exogenous DNA in the form of Donor homology plasmids or ODN (oligonucleotides) can be combined with the injection mix (containing sgRNA, Cas9 enzyme) so that the desired region can be incorporated into the specific site7,8. Furthermore, CRISPR/Cas9 DNA editing has the capability of targeting different genome edits in a single injection containing two different sgRNAs targeting multiple distinct regions which could be useful in reducing the overall time frame for complete editing10. Multiple bioinformatics tools are available online to design the sgRNA for the CRISPR system that will be helpful in assessing the maximum effectiveness before initiating the injection process.
Disadvantages of CRISPR
One major problem that researchers face when using the CRISPR/Cas9 gene-editing process is the increased probability of introducing DSBs in unintended regions which would be problematic in the downstream seeking for a precise edit10,11. These “off-target“ effects (i.e., sites that have a few nucleotide differences compared to the intended target) of genome editing might alter the gene function or genomic stability downstream hindering the probability of the direct application in the clinical procedure12. In general, CRISPR/Cas9 gene editing in zebrafish might be a time-consuming process in creating transmitting transgenic germline, in order to get more precise gene-editing and the possibility of INDELs (Insertions and Deletions) in the DNA sequence, we need to do thorough PCR screening and NGS (Next-generation sequencing) adding up the cost and time of the overall procedure.
Learn more about our Zebrafish Genome-Editing Services.
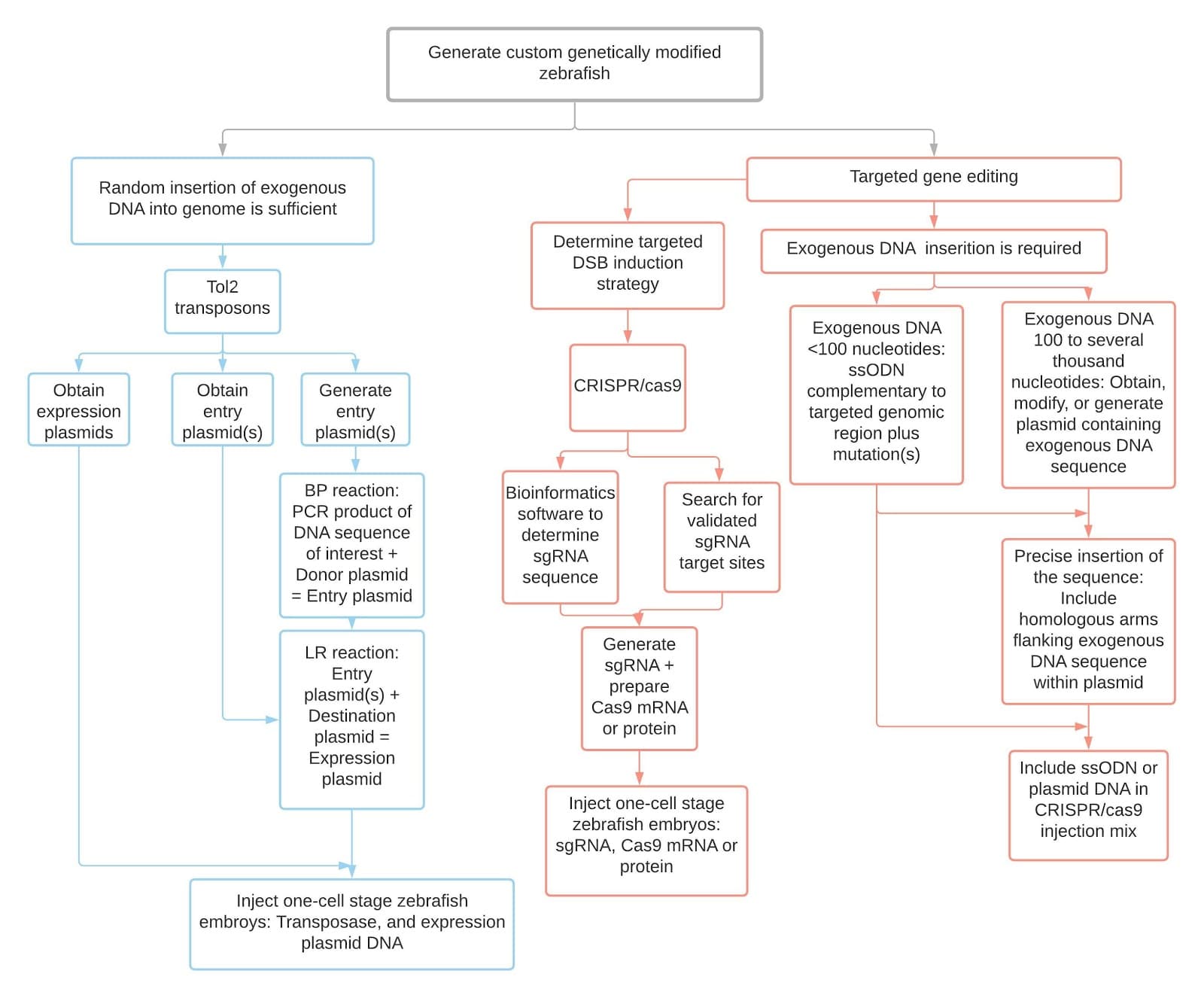
Figure 1: General zebrafish gene editing workflow indicating the tol2-transposons and CRISPR/cas9 methodology. The above flowchart is modified from the original article version5.
Tol2– transposons
Prior to the wide-use of CRISPR/Cas9-based tools on the genome editing of the zebrafish, Scientists had to rely on other methods such as Tol2 – transposons to modify the genome. The Tol2 transposon element was originally discovered in the genome of the medaka fish and its prevalence in other vertebrates like zebrafish made a prominent choice in creating transgenic zebrafish models14. After the transposon element discovery and its capability of foreign DNA integration from the plasmid into the genome of the fish, since then, it was widely adopted in zebrafish to insert cargo into the zebrafish germline to generate stable lines13,14. The technique relies first on the construction of plasmids which contain the matching DNA sequences in the same orientation and flanked by ITRs (inverted terminal repeats) specific to the Tol2 transposase5.
Plasmids can be generated by multiple molecular cloning methods, such as gateway cloning, Infusion cloning, and Restriction enzymes/T4 ligation methods14,15,17. Transposase mRNA is the preferred method of using the Tol2 injection mixture which could be obtained by reverse transcription of the linearized plasmid treated with restriction enzymes. In zebrafish, co-injection of mRNA encoding the transposase in the Tol2 injection mixture insures that the transposase is only available transiently and its integrations will be stable following natural mRNA degradation15. Tol2 transposase protein injections are also found to be effective in several research studies16. The initial steps involved in creating the transgenic zebrafish using Tol2 – mediated transposase is outlined in figure 2.
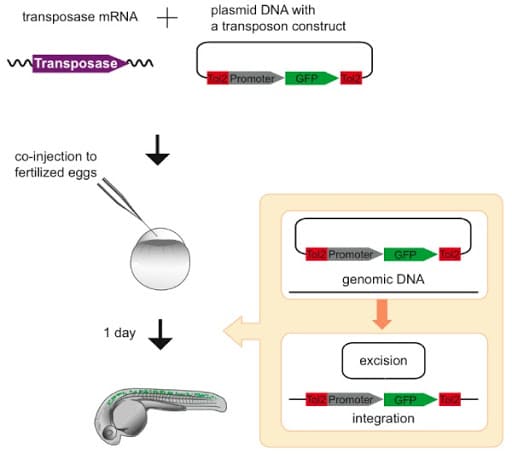
Figure 2: General overview of the Tol2-mediated transposase system depicting the initial steps involved in the Tol2- transposon injections. The above graphic is obtained from the published research article (Kawakami K. Tol2: a versatile gene transfer vector in vertebrates. Genome Biol. 2007;8 Suppl 1:S7-S7. doi: 10.1186/gb-2007-8-s1-s7).
Advantages of using Tol2- transposons
Tol2 is a powerful method of generating mutant zebrafish with random insertions. It is the most reliable method and has a higher efficiency in generating mutated offsprings than CRISPR/Cas9. Using Tol2 – transposase gene editing, it is possible to achieve 50-70% germline transmission efficiency to generate F1 transgenic offspring18,19. More importantly, transgene expression persists through generations, suggesting that transgenes created by Tol2-mediated transgenesis may not suffer from gene silencing effects31 creating long-term transgene expression33,34 in vertebrates.
Trangenesis constructed by DNA microinjection has a higher probability of carrying concatemers of the injected plasmid DNA in the organism genome28,29,30, which may lead to silencing of transgene expression after the passage through generations. On the contrary, integration of the Tol2 element does not cause any misarrangement of its surrounding genomic DNA, and transgenic fish with a single insertion can be easily isolated14. In addition to that, transgene expression in these fishes may not suffer from a gene-silencing effect. In fact, expression of GFP in transgenic fish created by Tol2‐mediated transgenesis persists after F5.
Furthermore, we are able to create a transgene that expresses Green fluorescence protein (GFP) using Tol2-transposase20,21. It is also much easier to handle and engineer Tol2 constructs22. Another major application of the Tol2 – mediated transient expression is its ability to conduct functional analysis of the non-coding sequences and cis-regulatory elements in the vertebrate genome22.
Although we could able to transfer a large cargo size of upto 10kb DNA sequences using Tol-2 transposon in Zebrafish32, lack of specificity of its genome integration of the Tol-2 cargo remains a concern making this technique unusable in precisely modifying specific genes or inserting transgenes into specific loci type scenarios. On the contrary, CRISPR/cas9 overcomes this limitation by precise gene editing, however CRISPR mechanisms are subjected to the relatively smaller cargo size. Several research studies concluded that the Tol2 transgenesis is able to mediate reliable gene expression which can persist through several generations under the control of regulatory elements22 but random insertion of the genetic sequences variants of the Tol-2 system is another limitation in zebrafish research5 which would be avoided certainly using CRISPR/cas9 systems.
Learn more about our Tol2 Transgenesis Services.
- Anzalone, A.V., Koblan, L.W. & Liu, D.R. Genome editing with CRISPR-Cas nucleases, base editors, transposases, and prime editors. Nat Biotechnol 38, 824-844 (2020). https://doi.org/10.1038/s41587-020-0561-9
- Press release: The Nobel Prize in Chemistry 2020. NobelPrize.org. Nobel Media AB 2020. Tue. 13 Oct 2020. <https://www.nobelprize.org/prizes/chemistry/2020/press-release/>
- Cornet, Carles et al. “Combining Zebrafish and CRISPR/Cas9: Toward a More Efficient Drug Discovery Pipeline.” Frontiers in pharmacology vol. 9 703. 3 Jul. 2018, DOI:10.3389/fphar.2018.00703
- Chang H. H. Y., Pannunzio N. R., Adachi N., Lieber M. R. (2017). Non-homologous DNA end joining and alternative pathways to double-strand break repair. Nat. Rev. Mol. Cell Biol. 18 495-506. 10.1038/nrm.2017.48
- Rafferty SA, Quinn TA. A beginner’s guide to understanding and implementing the genetic modification of zebrafish. Prog Biophys Mol Biol. 2018 Oct;138:3-19. DOI: 10.1016/j.pbiomolbio.2018.07.005. Epub 2018 Jul 19. PMID: 30032905.
- S. Albadri, F. Del Bene, C. Revenu Genome editing using CRISPR/Cas9-based knock-in approaches in zebrafish Methods, 121-122 (2017), pp. 77-85 https://doi.org/10.1016/j.ymeth.2017.03.005
- K. Hoshijima, M.J. Jurynec, D.J. Grunwald Precise genome editing by homologous recombination Meth. Cell Biol., 135 (2016), pp. 121-147 https://doi.org/10.1016/bs.mcb.2016.04.008
- H. Morita, K. Taimatsu, K. Yanagi, A. Kawahara Exogenous gene integration mediated by genome editing technologies in zebrafish Bioengineered, 8 (2017), pp. 287-295 https://doi.org/10.1080/21655979.2017.1300727
- Rafferty and Quinn, 2018 S.A. Rafferty, T.A. Quinn A beginner’s guide to understanding and implementing the genetic modification of zebrafish Prog. Biophys. Mol. Biol., 138 (2018), pp. 3-19
- P. Gut, S. Reischauer, D.Y.R. Stainier, R. Arnaout Little fish, big data: zebrafish as a model for cardiovascular and metabolic disease Physiol. Rev., 97 (2017), pp. 889-938 https://doi.org/10.1152/physrev.00038.2016
- J.D. Sander, J.K. Joung CRISPR-Cas systems for editing, regulating and targeting genomes Nat. Biotechnol., 32 (2014), pp. 347-350 https://doi.org/10.1038/nbt.2842
- Omodamilola OI, Ibrahim AU (2018) CRISPR Technology; Advantages, Limitations and Future Direction. J Biomed Pharm Sci 1: 115
- A. Koga, M. Suzuki, H. Inagaki, Y. Bessho, H. Hori Transposable element in fish Nature, 383 (30) (1996) https://doi.org/10.1038/383030a0
- K. Kawakami, A. Shima, N. Kawakami Identification of a functional transposase of the Tol2 element, an Ac-like element from the Japanese medaka fish, and its transposition in the zebrafish germ lineage Proc. Natl. Acad. Sci. Unit. States Am., 97 (2000), pp. 11403-11408 https://doi.org/10.1073/pnas.97.21.11403
- K.J. Clark, M.D. Urban, K.J. Skuster, S.C. Ekker Transgenic zebrafish using transposable elements Meth. Cell Biol., 104 (2011), pp. 137-149 https://doi.org/10.1016/B978-0-12-374814-0.00008-2
- J. Ni, K.J. Wangensteen, D. Nelsen, D. Balciunas, K.J. Skuster, M.D. Urban, S.C. Ekker Active recombinant Tol2 transposase for gene transfer and gene discovery applications Mobile DNA, 7 (2016) https://doi.org/10.1186/s13100-016-0062-z
- X. Chen, D. Gays, M.M. Santoro Transgenic zebrafish D. Ribatti (Ed.), Tumor Angiogenesis Assays: Methods and Protocols, Springer Science+Business Media (2016), pp. 107-114 https://doi.org/10.1007/978-1-4939-3999-2
- Urasaki A, Morvan G, Kawakami K. Functional dissection of the Tol2 transposable element identified the minimal cis-sequence and a highly repetitive sequence in the subterminal region essential for transposition. Genetics. 2006;174:639-649. doi: 10.1534/genetics.106.060244.
- Kawakami K, Takeda H, Kawakami N, Kobayashi M, Matsuda N, Mishina M. A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev Cell. 2004;7:133-144. doi: 10.1016/j.devcel.2004.06.005.
- Amsterdam A, Lin S, Hopkins N. The Aequorea victoria green fluorescent protein can be used as a reporter in live zebrafish embryos. Dev Biol. 1995;171:123-129. doi: 10.1006/dbio.1995.1265.
- Long Q, Meng A, Wang H, Jessen JR, Farrell MJ, Lin S. GATA-1 expression pattern can be recapitulated in living transgenic zebrafish using GFP reporter gene. Development. 1997;124:4105-4111.
- Kawakami K. Tol2: a versatile gene transfer vector in vertebrates. Genome Biol. 2007;8 Suppl 1:S7-S7. doi: 10.1186/gb-2007-8-s1-s7
- Howe, K., Clark, M. D., Torroja, C. F., Torrance, J., Berthelot, C., Muffato, M., et al. (2013). The zebrafish reference genome sequence and its relationship to the human genome. Nature 496, 498-503. doi: 10.1038/nature12111
- Dara, Mahintaj, and Mahdieh Talebzadeh. “CRISPR/Cas as a Potential Diagnosis Technique for COVID-19.” Avicenna journal of medical biotechnology vol. 12,3 (2020): 201-202.
- Hwang, W., Fu, Y., Reyon, D. et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31, 227-229 (2013). https://doi.org/10.1038/nbt.2501
- Moon, S.B., Kim, D.Y., Ko, JH. et al. Recent advances in the CRISPR genome editing tool set. Exp Mol Med 51, 1-11 (2019). https://doi.org/10.1038/s12276-019-0339-7
- Available at https://www.nature.com/subjects/cas9-endonuclease
- Stuart GW, McMurray JV, Westerfield M. 1988. Replication, integration and stable germ‐line transmission of foreign sequences injected into early zebrafish embryos. Development 103: 403-412.
- Culp P, Nusslein‐Volhard C, Hopkins N. 1991. High‐frequency germ‐line transmission of plasmid DNA sequences injected into fertilized zebrafish eggs. Proc Natl Acad Sci USA 88: 7953-7957.
- Cretekos CJ, Grunwald DJ. 1999. alyron, an insertional mutation affecting early neural crest development in zebrafish. Dev Biol 210: 322-338.
- Kawakami K. Tol2: a versatile gene transfer vector in vertebrates. Genome Biol. 2007;8 Suppl 1(Suppl 1):S7. doi:10.1186/gb-2007-8-s1-s7
- Balciunas D, Wangensteen KJ, Wilber A, Bell J, Geurts A, et al. (2006) Harnessing a High Cargo-Capacity Transposon for Genetic Applications in Vertebrates. PLOS Genetics 2(11): e169. https://doi.org/10.1371/journal.pgen.0020169
- Balciunas D., et al. (2006) Harnessing a high cargo-capacity transposon for genetic applications in vertebrates. PLoS Genet. 2, e169.
- Tsukahara T., et al. (2015) The Tol2 transposon system mediates the genetic engineering of T-cells with CD19-specific chimeric antigen receptors for B-cell malignancies. Gene Ther. 22, 209-215