C. elegans as a model to evaluate the function of disease genes
In 1998, the soil nematode Caenorhabditis elegans became the first multicellular organism of which the genome has been sequenced completely [1]. One surprising result of this approach was that ~65% of the human disease genes have a counterpart in the worm [2].
In recent years C. elegans have been established as a superb model to evaluate the function of disease genes. They suggest that indeed the large degree of sequence similarity found in genes in C. elegans and human genomes results in a significant functional similarity of the encoded proteins. Most importantly, the major signaling pathways of C. elegans and vertebrates, including Ras, Notch, TGFβ, wnt/wingless and insulin signaling are remarkably conserved [3].
In addition to the ease of genetic manipulations, this tiny nematode has one major advantage that might develop it into a superb tool for the biotech industry – C. elegans can easily be grown in microtiter plates in both 96- and 384-well formats. Because its receptor pharmacology is remarkably similar to that of humans, drugs and side effects can be tested in large scale using C. elegans and its mutants as an in vivo model.
 Recently, considerable effort has been made to study C. elegans genes whose mutations in the human counterparts result in the onset of complex neurological disorders, such as Alzheimer’s, Huntington’s and Parkinson’s diseases.
Recently, considerable effort has been made to study C. elegans genes whose mutations in the human counterparts result in the onset of complex neurological disorders, such as Alzheimer’s, Huntington’s and Parkinson’s diseases.
Learn more about C. elegans genes associated with high-impact human diseases.
Our team successfully inserted the human coding sequence of STXBP1 into the native locus of a C. elegans ortholog gene. We call the technique a “Gene-Swap” because the worms version of the gene is replaced with a human coding sequence. The Gene-Swapped STXBP1 sequence functioned and gave a high level of rescue activity (the “Humanized” strain). A knock-out was made by precisely deleting the unc-18 locus (ortholog of STXBP1). In the process, all coding sequence of unc-18 is removed and a full loss-of-function deletion allele is generated. In the gene variant we insert the p.Arg406His into the humanized locus.
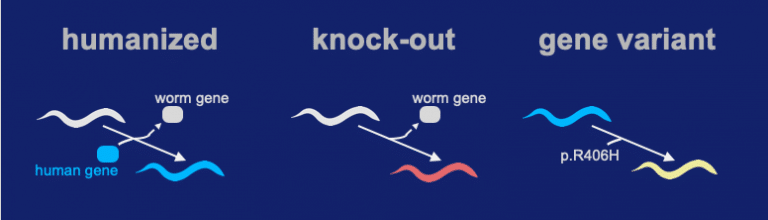
The gene-swap humanized strain for expressing hSTXBP1 shows a significant level of activity (see video below). In contrast, the knock-out shows very little activity. For the R406H genomic variant, its activity is somewhere in between the “humanized” and “knock-out” strain.
Behavior quantified using three assays
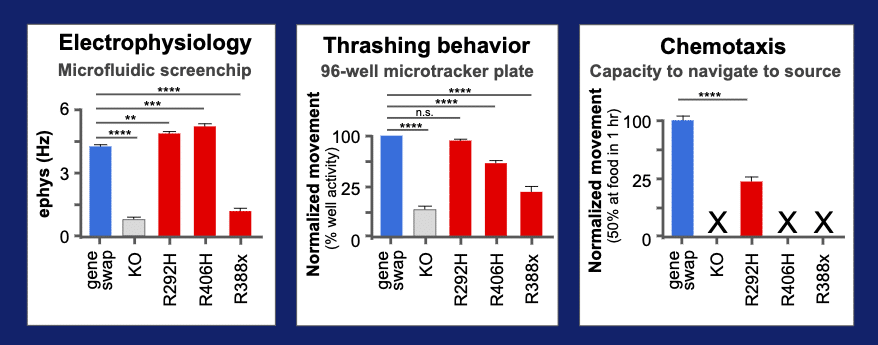
R406H’s deviant behavior was quantified in a microfluidics system that measures an EEG-like electrical activity of the animal. We added two other assays on this variant – a wMicroTracker test for thrashing in liquid and and a chemotaxis test for speed to navigate to a food source. We also added two other reference alleles that are also known to be pathogenic variants of STXBP1 (R292H and R388X). The three variants in red can be compared to the wild type “gene-swap” for the humanized line in blue. In the elecrophysiology assays, we can see that two variants, R292H and R406H, have increased rate of neurotransmission. In contrast, the R388X allele has a lower rate of transmission. Adding in the thrashing assays for movement in liquid, we see that the R292H has lost its deviant behavior, while the R406H and R338X have retained a detectable level of altered function. A final assays was performed that uses a sensitive chemotaxis behavioral assay. This assay measure the ability of animal to reach a food source in 1 hr. All variants show altered function and only the R292H allele shows residual level of activity, so we can see that a series of assays measure behavioral differences and give a glimpse into the mechanism of disease.
‘Humanized worms’ can now be used to identify modifier genes that might define the cellular mechanisms these factors are involved in. Look out for more examples of functional conservation, perhaps even of entire pathways, in the near future!
References:
1. The C. elegans Sequencing Consortium. (1998) Genome sequence of the nematode C. elegans: a platform for investigating biology. Science 282, 2012–2018
2. Sonnhammer, E.L. and Durbin, R. (1997) Analysis of protein domain families in Caenorhabditis elegans. Genomics 46, 200–216
3. The worm in us – Caenorhabditis elegans as a model of human disease. TRENDS in Biotechnology Vol.20 No.4 April 2002, p.147.

