Summary
The nematode C. elegans has been used as a model organism for nearly sixty years and has made significant contributions to science in that time. In this white paper we will discuss the attributes of C. elegans, their advantages and limitations, and how they are currently being used in research to answer the question: is a C. elegans model right for my research?
Download this free white paper to learn more about the advantages and limitations of C. elegans models, the scientific discoveries they’ve contributed to, and the research areas they are widely used in.
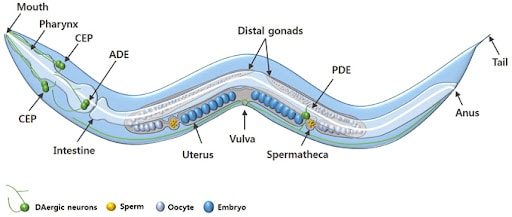
Figure 1. An adult C. elegans hermaphrodite. The DAergic neurons include 4 cephalic (CEP) neurons, 2 anterior deirid (ADE) neurons, and 2 posterior deirid (PDE) neurons. Males have 6 additional DAergic neurons located in the tail (Chege & McColl, 2014)
What is a C. elegans?: Biological Sketch
C. elegans is a free-living nematode, a member of the phylum Nematoda [See Figure 1]. A roundworm, C.elegans is a relatively small organism, growing to 1mm, which mainly consists of their reproductive system (Edgley & the Riddle Lab, 2015). There are two sexes: male or hermaphrodite, but they are easily differentiated by the shape of their tails [Figure 2] (Yokoyama, 2020; Meneely, Dahlberg & Rose, 2019). Its body has three layers: an epidermal, a muscular, and an intestinal layer.
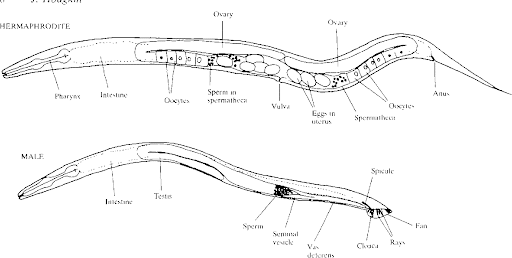
Figure 2. Primary sex determination in the nematode C. elegans (Hodgkin, 1987).
Origins of the Model: Out of all the worms,
why C. elegans?
When asking ‘why are C. elegans widely used as a model organism instead of another type of nematode (worm)’, the credit belongs to Sydney Brenner. Sydney Brenner was a genetic biologist who had the foresight to specifically choose C. elegans because it was well-suited to genetic analysis – his work on this model would lead him to receive a Nobel Prize in Physiology or Medicine in 2002 (Brenner, 2002). Brenner first started thinking about the model in 1963 when he wrote to the chairman of the Medical Research Council’s Laboratory of Molecular Biology (MRC-LMB) outlining the future of molecular biology research. In this letter Brenner actually suggested the nematode Caenorhabditis briggsae as the model organism to shepherd in the next decade of discoveries, however, Brenner later changed his mind, settling on the Caenorhabditis elegans in 1965 because it grew better in the lab (Corsi, Wightman & Chalfie, 2015; Riddle, Blumenthal, Meyer & Priess, 1997).
Advantages to using the C. elegans Model
C. elegans is an ideal laboratory organism because it is inexpensive, requiring low maintenance, and little space. Only growing to ~1mm, C. elegans can be grown in petri dishes, stored in large numbers in the lab, and don’t require many other resources commonly needed for other organisms, such as special diets or environmental enrichment that is needed for mammals.(Corsi., Wightman & Chalfie, 2015). Their short life cycle (2-3 weeks) also enables lifespan studies to be conducted much faster than traditional mammalian models [Figure 3]. Furthermore, because of their hermaphroditic nature, C. elegans can self-fertilize, enabling them to reproduce quickly in high numbers ( ∼300 offspring, with a reproductive cycle of 3.5 days) (Meneely, Dahlberg & Rose, 2019). C. elegans have the added advantage of withstanding a ”starved’ state for months or, for longer storage periods, they can be frozen at -80C and later revived. Their ability for cryopreservation is especially beneficial for genetic research, as mutant strains can be preserved without the concern that background mutations, suppressors, or other modifiers will occur (Corsi., Wightman & Chalfie, 2015).
Another advantage of C. elegans is that, unlike mammalian models which have to adhere to numerous animal testing regulations, C. elegans have few ethical concerns (Zhang, et al., 2020). An early criticism of C. elegans was that they were too simplistic to be used as an alternative model to these mammalian models, however, in 1974 Brenner published the first genetic map of the organism, revealing that the C. elegans has over 100 genetic loci dispersed over 6 chromosomes, all of which are behavioural or morphological markers (Corsi., Wightman & Chalfie, 2015). This publication made it apparent that C. elegans were particularly powerful for genetic research, as their simplistic qualities lend themselves to research on complex genetics: their transparent body enables observation at the single-cell level. Additionally, in 1998 they became the first organism to have their genome fully sequenced (C. elegans Sequencing Consortium, 1998). Thus, C. elegans are extremely advantageous for developmental studies (Corsi., Wightman & Chalfie, 2015). C. elegans’ transparent body also enables the use of fluorescent protein reporters, such as Cameleon and gCaMP3 that allow researchers to measure electrophysiology in vivo (Corsi., Wightman & Chalfie, 2015).
Despite C. elegans being a simplistic model there are many structural overlaps between humans and C. elegans such as the: digestive system, nervous system, and reproductive system. C. elegans also display complex behaviour and have ~65% of disease genes in common with humans (Baumeister & Ge, 2002). Taken together, these characteristics make C.elegans capable of a wide range of studies, and able to produce robust and translatable data.
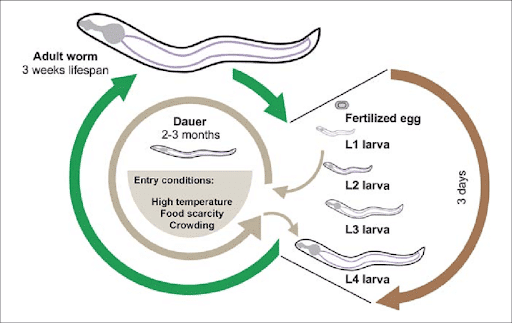
Figure 3. The lifecycle of the C. elegans (Ewald, Castillo-Quan & Blackwell, 2017).
From a practical standpoint, C. elegans are also an advantageous model because they are easy to work with without the hindrances to researchers that other models can pose: there is little health risk as the worm cannot bite, scratch, or grow at body temperature (cannot grow in humans), and there are no recorded allergic reactions (Corsi., Wightman & Chalfie, 2015). Moreover, the active, and collaborative, C. elegans community means there are vast resources available for C. elegans researchers. These include databases such as: WormBase (https://wormbase.org), which houses nearly all of the known genetic and genomic information of C. elegans, the Caenorhabditis Genetics Center (http://cgc.umn.edu), which distributes strains and has information on gene names and laboratory contacts, and WormBook (https://wormbook.org), which collects up-to-date articles on C. elegans discoveries and laboratory practices. The access to a complete cell lineage map, these knockout (KO) mutant libraries, and established genetic methodologies including mutagenesis, transgenesis, and RNA interference (RNAi) provide a variety of options to manipulate and study C. elegans at the molecular level (Leung et al., 2008).
C. elegans has always been collaborative, so I do love that part of the worm community.”
– Dr Anne Hart, 17 Minutes of Science
Limitations to the C. elegans Model:
Like any model, C. elegans has its limitations. Mainly, their simple body plan, and lack of certain human anatomical features such as bones, blood, a defined fat cell, and internal organs (heart or circulatory system) make them unable to model certain conditions. Similarly, for certain research, C. elegans’ small size may also prove challenging (Tissenbaum, 2015).
Awards C. elegans have contributed to: six Nobel Prizes
As previously mentioned, C. elegans was used in Sydney Brenner’s work on organ development and programmed cell death, for which he, John E. Sulston, and H. Robert Horvitz shared the 2002 Nobel prize. C. elegans also enabled the Nobel-winning work of three other researchers: Andrew Z. Fire and Craig C. Mello’s discovery of the phenomenon “RNA interference” (2006), and Martin Chalfie’s discovery and development of the Green Fluorescent Protein (GFP) (2008) (Nobel Prize Outreach, 2021; Chalfie, 2008). Thus, despite only gaining prominence as a model organism a few decades ago, Brenner rightly foresaw C. elegans’ power as a model.
Research Areas C. elegans are widely/regularly used in:
C. elegans & the Nervous System: Recent & Notable Publications
- C. elegans as a model to study glial development
- The Use and Predictability of C. elegans as an Alternative and Complementary Model in Neurotoxicological Studies: Focus on the Dopaminergic System
- What about the males? the C. elegans sexually dimorphic nervous system and a CRISPR-based tool to study males in a hermaphroditic species
C. elegans & the Immune System : Recent & Notable Publications
- Neuronal GPCR NMUR-1 regulates distinct immune responses to different pathogens
- Interspecies bacterial competition regulates community assembly in the C. elegans intestine
- The Microbial Zoo in the C. elegans Intestine: Bacteria, Fungi and Viruses
C. elegans & Rare Disease Research : Recent & Notable Publications
C. elegans & Aging / Longevity
- Caenorhabditis elegans as a model system for studying aging-associated neurodegenerative diseases
- Age-dependent changes and biomarkers of aging in Caenorhabditis elegans
- Lifespan Extension of Caenorhabditis elegans by Butyricicoccus pullicaecorum and Megasphaera elsdenii with Probiotic Potential
C. elegans & Toxicology Research
- Caenorhabditis elegans: An Emerging Model in Biomedical and Environmental Toxicology
- Caenorhabditis elegans As a Promising Alternative Model for Environmental Chemical Mixture Effect Assessment-A Comparative Study
- The C. elegans model in toxicity testing
- Caenorhabditis elegans for predictive toxicology
Download this free white paper to learn more about the advantages and limitations of C. elegans models, the scientific discoveries they’ve contributed to, and the research areas they are widely used in.
With its low health risk and absence of recorded allergic reactions, C. elegans is a practical and advantageous model for many types of research. And with the active and collaborative C. elegans community, there are vast resources available to support your research. At InVivo Biosystems, we offer a C. elegans custom injection service to help you create the perfect transgenic worm strain for your experiments.
References
- Baumeister, R., & Ge, L. (2002). The worm in us – Caenorhabditis elegans as a model of human disease. Trends in biotechnology, 20(4), 147-148. https://doi.org/10.1016/s0167-7799(01)01925-4
- Brenner, Sydney (2002). Nobel Lecture. NobelPrize.org. Nobel Prize Outreach AB. <https://www.nobelprize.org/prizes/medicine/2002/brenner/lecture/>
- C. elegans Sequencing Consortium (1998). Genome sequence of the nematode C. elegans: a platform for investigating biology. Science (New York, N.Y.), 282(5396), 2012-2018. https://doi.org/10.1126/science.282.5396.2012
- Chalfie, Martin (2008). Nobel Lecture. NobelPrize.org. Nobel Prize Outreach AB. https://www.nobelprize.org/prizes/chemistry/2008/chalfie/lecture/
- Chege, P. M., & McColl, G. (2014). Caenorhabditis elegans: a model to investigate oxidative stress and metal dyshomeostasis in Parkinson’s disease. Frontiers in aging neuroscience, 6, 89. https://doi.org/10.3389/fnagi.2014.00089
- Corsi A.K., Wightman B. & Chalfie M. A (2015). Transparent window into biology: A primer on Caenorhabditis elegans, WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.177.1, http://www.wormbook.org.
- Edgley & the Riddle Lab (2015). WHAT IS CAENORHABDITIS ELEGANS AND WHY WORK ON IT? University of Minnesota, College of Biological Sciences. https://cbs.umn.edu/cgc/what-c-elegans
- Ewald, Collin Y., Castillo-Quan, Jorge I. & Blackwell, Keith, T. (2017) Untangling Longevity, Dauer, and Healthspan in Caenorhabditis elegans Insulin/IGF-1-Signalling. Gerontology; 64:96-104. https://doi.org/10.1159/000480504
- Hodgkin, J. (1987). Primary sex determination in the nematode C. elegans. Development, 101 Suppl, 5-16 .
- Leung, Maxwell C. K.,Williams, Phillip L., Benedetto, Alexandre Au, Catherine,Helmcke, Kirsten J., Aschner, Michael, Meyer, Joel N. (2008) Caenorhabditis elegans: An Emerging Model in Biomedical and Environmental Toxicology, Toxicological Sciences, 106 (1), 5-28, https://doi.org/10.1093/toxsci/kfn121
- Meneely, P. M., Dahlberg, C. L., & Rose, J. K. (2019). Working with worms: Caenorhabditis elegans as a model organism. Current Protocols Essential Laboratory Techniques, 19, e35. doi: 10.1002/cpet.35
- Nobel Prize Outreach (2021). Andrew Z. Fire – Facts. NobelPrize.org, https://www.nobelprize.org/prizes/medicine/2006/fire/facts/
- Riddle DL, Blumenthal T, Meyer BJ, & Priess, JR. editors. (1997). C. elegans II. 2nd edition. Cold Spring Harbor Mongraph Series, Vol. 33. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press; available from: https://www.ncbi.nlm.nih.gov/books/NBK19997/
- Tissenbaum H. A. (2015). Using C. elegans for aging research. Invertebrate reproduction & development, 59(sup1), 59-63. https://doi.org/10.1080/07924259.2014.940470
- Yokoyama, Maho. (2020). C. elegans as a Model Organism. News-Medical.Net, https://www.news-medical.net/life-sciences/C-elegans-as-a-Model-Organism.aspx
- Zhang, S., Li, F., Zhou, T., Wang, G., & Li, Z. (2020). Caenorhabditis elegans as a Useful Model for Studying Aging Mutations. Frontiers in endocrinology, 11, 554994. https://doi.org/10.3389/fendo.2020.554994
InVivo Biosystems’ experienced team can take over and deliver the results you need through our C. elegans gene editing services. We welcome the opportunity to discuss your project with you.




