The Role of C. elegans in Lifespan Research
In the past 20 years, research done with the nematode model, C. elegans, has been pivotal in revealing the underlying biological mechanisms of aging in humans. C. elegans is a uniquely situated model organism for studying lifespan and aging, particularly through compound-mediated longevity experiments. With its short generation time, well-documented developmental genomics, and the extensive availability of modern assays and genetic sequencing techniques, C. elegans longevity experiments provide key details into how different compounds affect lifespan, healthspan, and reveal underlying transcriptional markers of health. Government institutions and private labs across the world are still using C. elegans to test different lifespan and healthspan interventions, including one effort known as the Caehornabitis Intervention Testing Program (CITP), a partnership between the National Institute of Aging and National Institute of Health.
Innovations in Lifespan Analysis
For decades, lifespan assays in C. elegans have been performed manually by researchers and their teams, often requiring enormous amounts of hands-on time and a 7 day work week. Technological upgrades have been advancing all industries, so why were lifespan assays still requiring so much manual labor? We knew there had to be a better, more efficient way to perform lifespan assays so we developed the Longevity Platform. Our Longevity Platform utilizes an automated lifespan assay developed in conjunction with leading academic partners. Our software tracks the worms, their movements, and provides critical data to the researchers in order to determine lifespan curves all without the need for human intervention past the early development stage of the C. elegans life cycle.
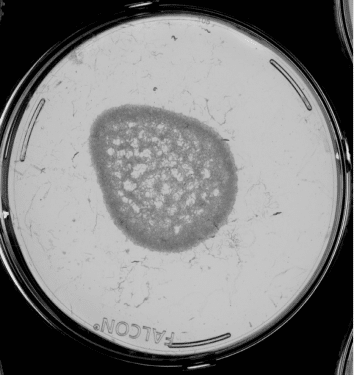

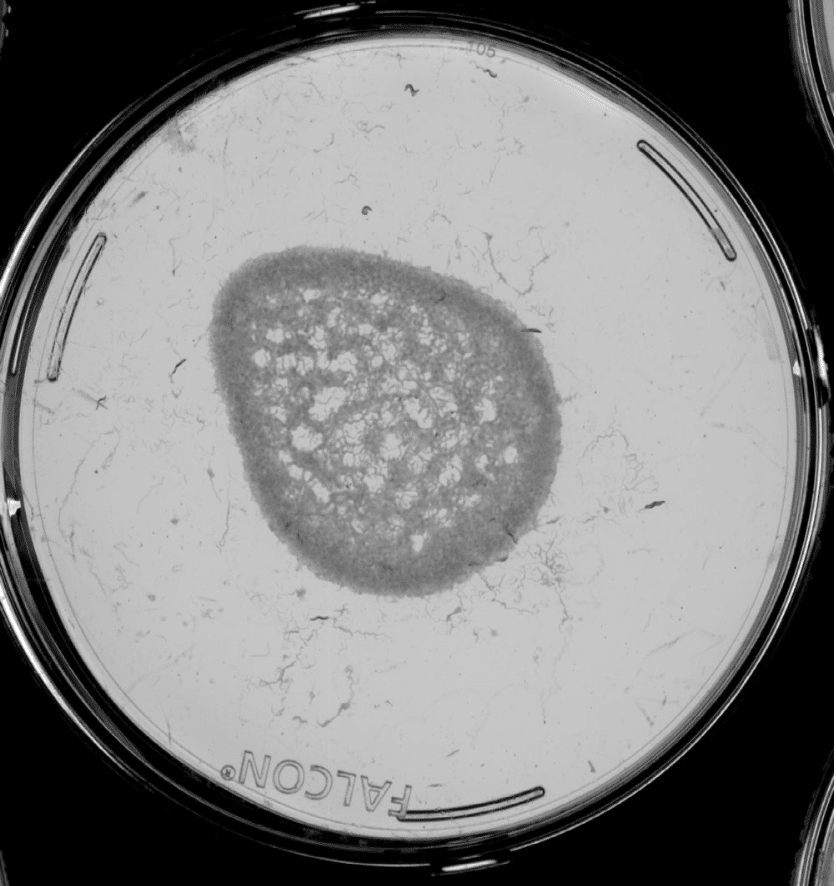
Images from the Longevity Platform. Left: active bacteria; Right: Inactive bacteria.
The Challenge of Reproducibility
Designing this automated platform forced us to reconsider some of the small details of lifespan assays. One such detail we had to consider was food source, a topic with many variations in the literature. Typical C. elegans studies use E. coli, such as the common lab strain OP50, to feed worms; but during development of the automated assay, we found that live OP50 caused a lot of variability in results. We opted instead to try a specialized strain of bacteria that can be “UV Inactivated.” Changing the bacterial strain that is fed to the worms may seem like a small, trivial detail, but it was absolutely essential for us. By doing so, we ensured our lifespan assays will always produce data reflecting the true influence a compound might have on extending (or shortening) lifespan in C. elegans.
What is UV Inactivated Bacteria?
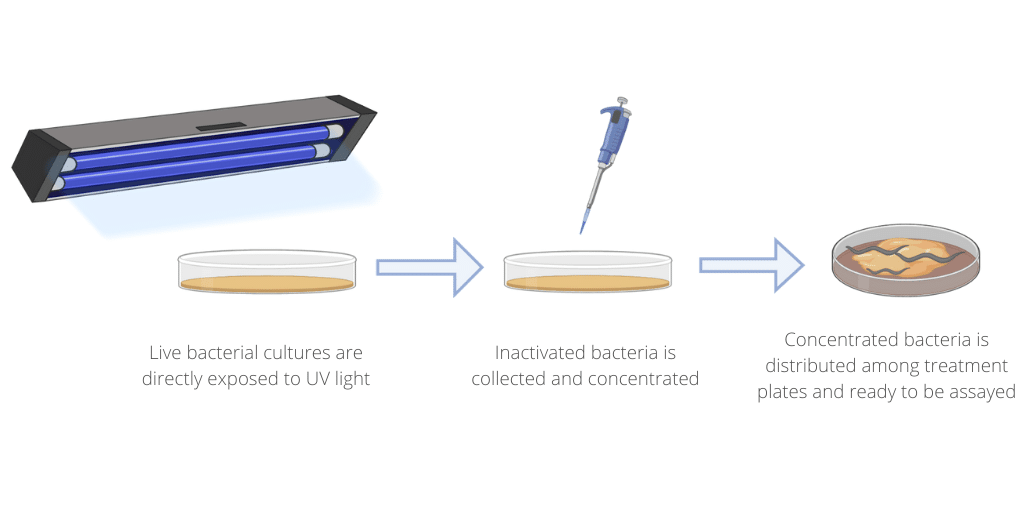 The term “inactivated bacteria”, in this context, is synonymous with “dead bacteria.” UV-inactivated bacteria is when that bacteria is “inactivated” (i.e. killed) by exposure to UV radiation. UV light exposure, be it from the sun or a UV light bulb, causes damage to the genetic code of cells leading to mutations that can oftentimes be lethal for those exposed cells. Over time, mechanisms of repair for UV damage to genes have developed in a wide range of living organisms, and bacteria such as E. coli is no exception.
The term “inactivated bacteria”, in this context, is synonymous with “dead bacteria.” UV-inactivated bacteria is when that bacteria is “inactivated” (i.e. killed) by exposure to UV radiation. UV light exposure, be it from the sun or a UV light bulb, causes damage to the genetic code of cells leading to mutations that can oftentimes be lethal for those exposed cells. Over time, mechanisms of repair for UV damage to genes have developed in a wide range of living organisms, and bacteria such as E. coli is no exception.
To account for this, specialized strains of laboratory E. coli were developed where the repair mechanisms were rendered ineffective. The purpose of this was to have cultures of bacteria that were particularly susceptible to UV radiation-mediated damage. The bacteria were exposed to a high dose of UV light (about the same amount of energy to melt 1g of ice) in a shallow petri dish before being spun down to the appropriate concentration for the lifespan assay. From there, the bacteria was transferred onto agar plates and allowed to fully dry before the worms were introduced.
Why is UV Inactivated Bacteria so Important?
What makes UV Inactivation essential to achieving reproducible and trustworthy data from the Longevity Platform? The answer boils down to a key facet of working with living organisms: the biotransformation of molecules! As you know, every day your body takes in different compounds/molecules and transforms them into a new form that is more suitable to your body’s metabolic needs, while extruding the leftovers in a… less than pleasant fashion.
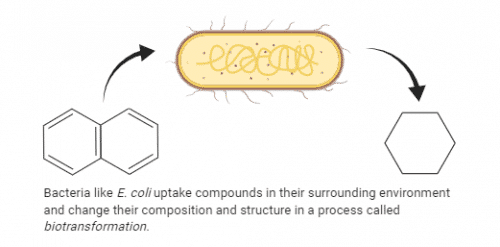
Bacteria such as E. coli are no strangers to this process. They uptake a variety of compounds in their vicinity and metabolize them into a different substance before excreting them back into their environment as something new, a process known as biotransformation. This presents a major issue when investigating the effects of a compound on the lifespan of C. elegans: what if the bacteria is biotransforming the compound of interest into something different? This could lead to skewed and/or inconsistent results.
While the biotransformation of the compound of interest by the bacterial food source for the worms was our main concern, the adoption of UV inactivated bacteria also provided two other ancillary benefits. When live bacteria is used to “seed” a plate and create a lawn of food for the worms to grow in, that bacteria does not necessarily stop growing once it is distributed on an agar plate. If left to grow longer, any discrepancies in the volume of bacteria added to an agar plate will lead to differences in the total amount of bacteria on the plate.
When testing the lifespan of an organism, it’s reservoir of available food is a key determining factor in the length of that lifespan, so having a consistent amount of bacteria in the lawn on each and every plate involved in the assay is essential to achieving clean, tight, reproducible results. When bacteria is left to grow on an agar plate, the bacterial lawn tends to form a pronounced ‘ridge’ around the edge of the area the lawn is covering. If any worms were to sit and hang out in the thick ridge of the bacterial lawn, we risk not being able to detect them with our worm-tracking software, which would nullify a large portion of the data generated.
Challenging, but Rewarding
Lifespan assays are a complicated technique. Automated lifespan assays are even more so. It is important to take stock of every decision, every aspect, and every angle in the development of these assays, because something as simple as “is the bacteria alive or dead” can drastically affect the outcome of your experiment. The Longevity Platform we provide at InVivo Biosystems has been meticulously vetted for these aspects in order to ensure the most accurate and representative data is generated when testing the effectiveness of a compound to extend or shorten lifespan.

About the Author: Tanner Feustel
Tanner is an R&D Laboratory Technician working with C. elegans, primarily on the Longevity Platform. He received his Bachelor of Science from the University of California, Davis in Animal Sciences with an emphasis on Genetics in 2018. Tanner joined the InVivo Biosystems team in May of 2019 and since then has been a part of multiple team projects, the most recent of which being the development of the Longevity Platform. Outside of work, he is passionate about science outreach, wildlife rehabilitation, and his favorite baseball team, the San Francisco Giants.